Endoscopic repair of CSF leak
1. Introduction
Traumatic CSF rhinorrhea occurring in the anterior and/or middle cranial fossa can be treated by endoscopic techniques associated with lumbar intrathecal administration of fluorescein.
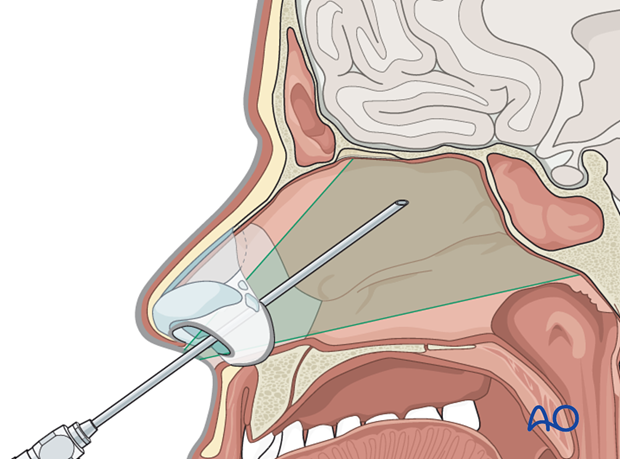
The illustration shows the region that can be reached endoscopically.
2. Tools required
Tool required for the endonasal endoscopic repair are:
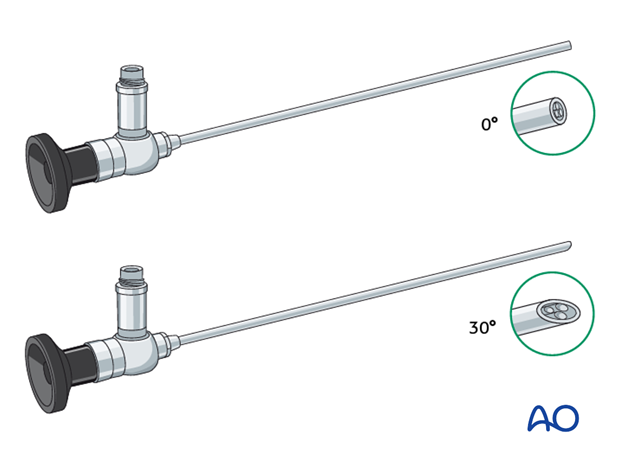
- Endoscopic set (0°, 30°, and 45° endoscopes)
- Fluorescein blue-light filter system coupled to the light source
- Fluorescein
3. Sequence of procedure
The steps for the surgical procedure are:
- Lumbar intrathecal fluorescein injection
- Endonasal endoscopy and identification of the defect
- Repair of the defect
4. Fluorescein injection
In the majority of cases before surgery a lumbar drainage is inserted which allows the injection of 1 ml sodium fluorescein (0.5%). Using a lumbar drainage offers the advantage to re-inject fluorescein during the surgical procedure if needed.
5. Endonasal endoscopy and identification of the defect
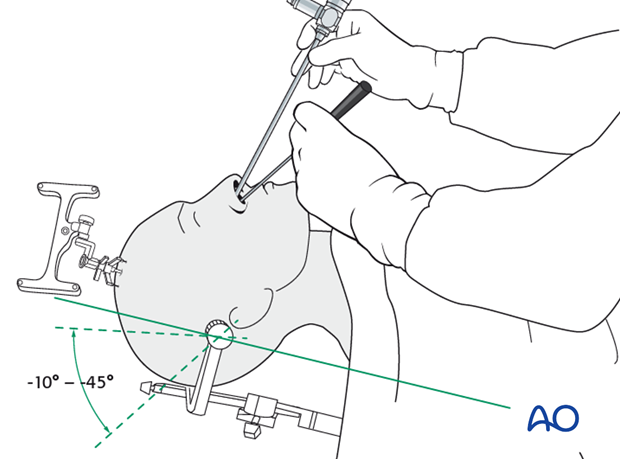
The patient can be positioned supine on the operating table with the trunk raised between -10° up to +45°. The head can be rotated toward the surgeon if necessary. The endoscope is inserted in the nostril (right, left, or both) and the anatomical landmarks are visualized.
The use of navigational devices (fusion data sets, CT, and MRI) is helpful.
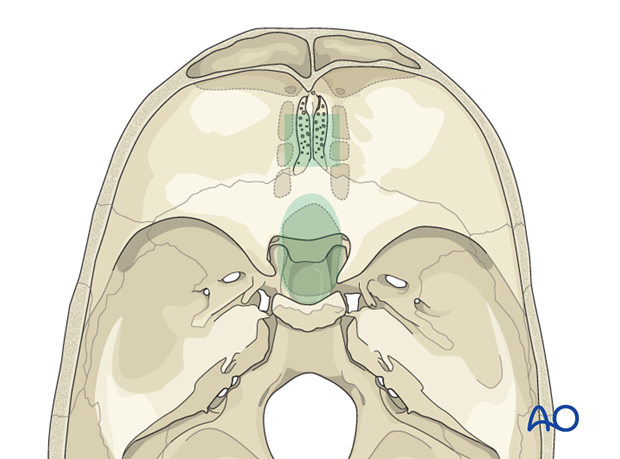
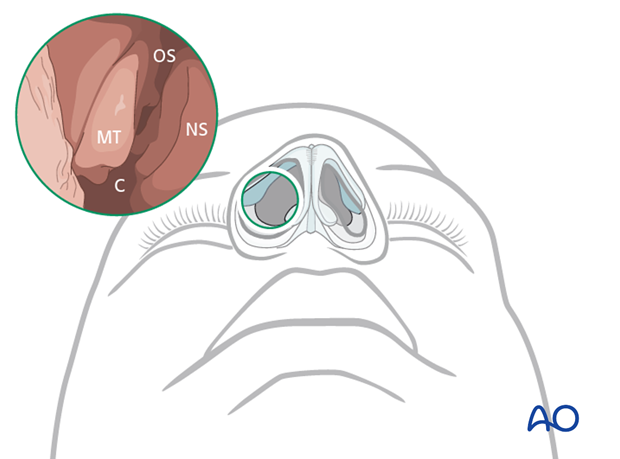
6. Identification of the anatomical landmarks
For the endoscopic anatomical orientation the following structures should be identified:
- Nasal septum (NS)
- Middle turbinate (MT)
- Choana (C)
- Sphenoid ostium (OS)
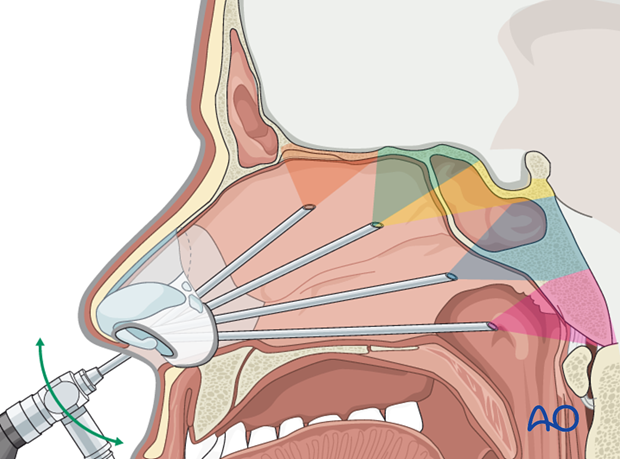
7. Methods of approach
Endonasal endoscopy can be performed by several methods depending on the type of lesion and its location. The classic approach to the olfactory fossa is the direct paraseptal one (with or without removing any ethmoidal structures according to the anatomical variations of the patient).
8. Identification of the lesion
The use of a fluorescein blue-light filter system and the fluorescein barrier filter mounted to the eyepiece of the endoscope might be helpful to visualize the localization of the dural defect. A CSF fistula, if present, is seen with a characteristic green glow.
Video showing endoscopic identification of a CSF leak using fluorescein
9. Repair of the defect
Several techniques exist for the repair of the defect and different grafts can be used including autologous nasal, extra-nasal, and heterologous grafts. The bone defects can be repaired using septal cartilages, parts from the middle nasal turbinates, eventually the vomer, etc. The most used autologous extranasal graft is the abdominal fat or the fascia lata. In larger defects, a vascularized nasoseptal flap can be used.
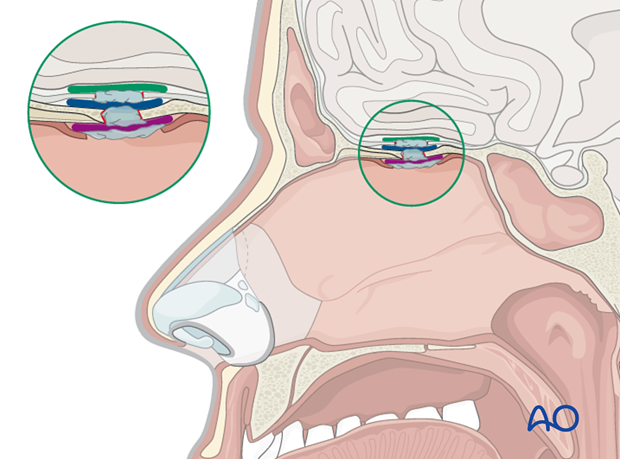
The closure technique is a strictly related to the individual patient’s anatomy, the size of the leak, and its anatomical location. Underlay, overlay, combined, and obliterative techniques have been described.
The illustration shows a combined three layer technique in which are evident:
- Subdural intracranial underlay graft (dark green)
- Extradural intracranial underlay graft (blue)
- Extracranial overlay graft (purple)
Fibrin glue can be used to keep the layers together or to fill the dead-space.
10. Confirmation of proper closure
After closing the defect, its efficacy and reliability is checked using a Valsalva maneuver and in special cases, intraoperative fluorescein test.
11. Aftercare following endoscopic management of skull base fractures
General postoperative care
- Postoperatively, a period of a supine bed rest (1-2 days) is necessary with the trunk and head raised 30°.
- Use antibiotics in the perioperative period.
- Patient follow-up after discharge. The patient is seen 4 weeks and then every 3-6 month in the first 2 postoperative years.













