Innervated free muscle transfer (Gracilis)
1. Introduction
Gracilis free flap transfer is the preferred option for facial reanimation for patients with irreversible or long-standing facial paralysis.
It offers the best chance of obtaining facial symmetry, voluntary movement, and natural appearing smile.
2. Technique
This operation is most efficiently performed using the two-team approach. The preparation of the face and the gracilis flap harvest can then be performed simultaneously.
Preparation of recipient site (face)
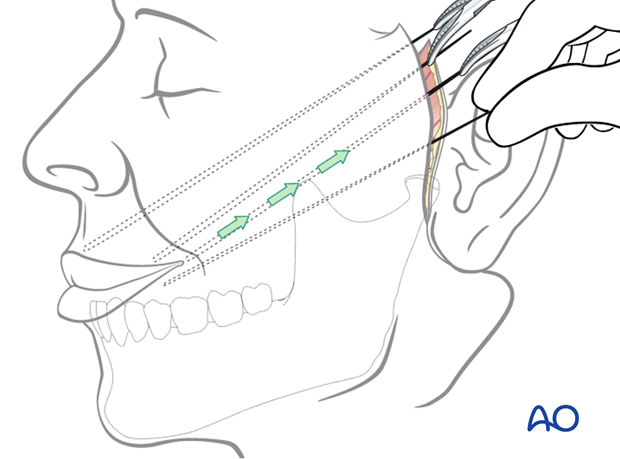
A parotidectomy type incision is performed.
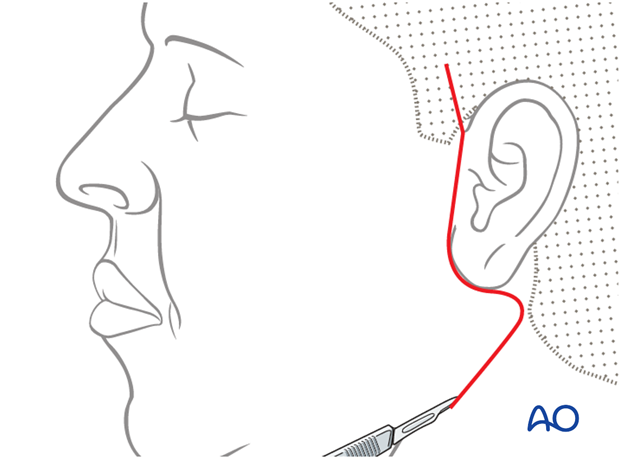
Elevation of the skin flap is performed over the superficial musculo-aponeurotic system (SMAS) fascia. The extent of dissection of the skin flap is taken to the oral commissure and beyond the nasolabial fold on the upper lip.

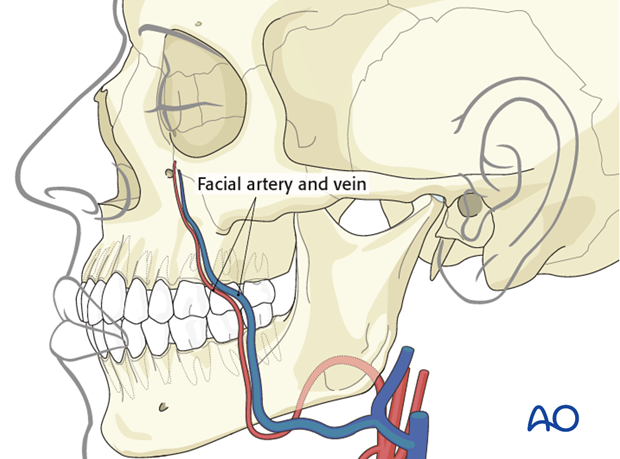
Recipient vessels
The facial artery and vein are most commonly used as the recipient vessels.
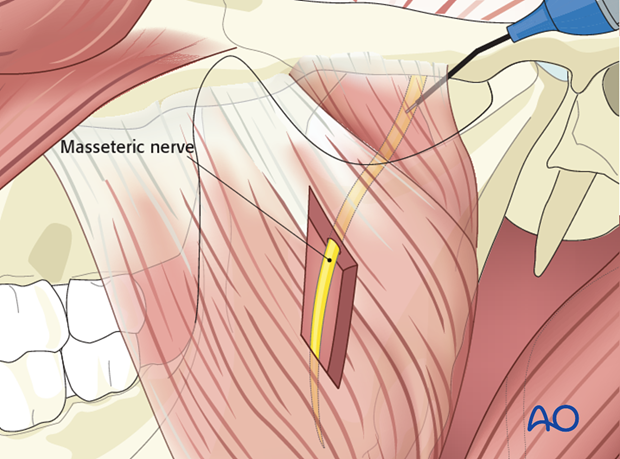
Recipient nerves
The nerve to masseter is commonly used to power the gracilis.
Other nerve options include: cross-facial nerve graft or the ipsilateral proximal facial nerve, when available.
The advantages of the masseteric nerve include: high motor neuron input and close proximity to the gracilis muscle (allowing direct nerve coaptation). This allows early, reliable, and strong reinnervation.
It is possible to use a combination of contralateral/ipsilateral facial nerve and masseteric nerve, in order to obtain strength of movement and spontaneity.
The masseteric nerve is identified within the body of the masseter muscle at the level of the sigmoid notch.
A nerve stimulator is used to confirm activity prior to nerve division and preparation for coaptation.
Preparation of donor site (gracilis muscle)
Incision and exposure of the gracilis muscle is performed in the standard fashion.
Ipsilateral gracilis is most commonly used, to allow for correct orientation of the neurovascular pedicle.
The harvest technique of the gracilis is beyond the scope of this module and is described elsewhere.
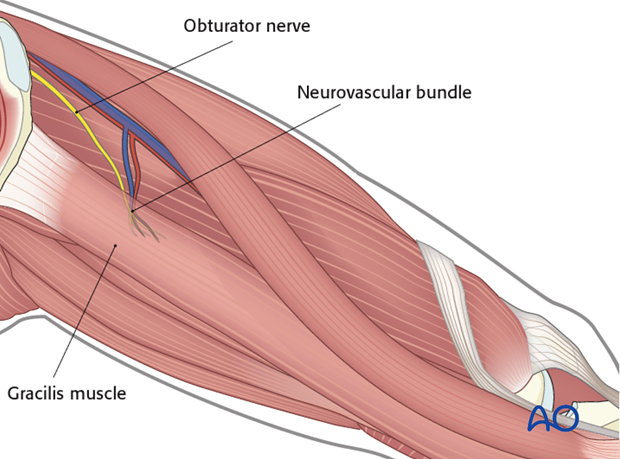
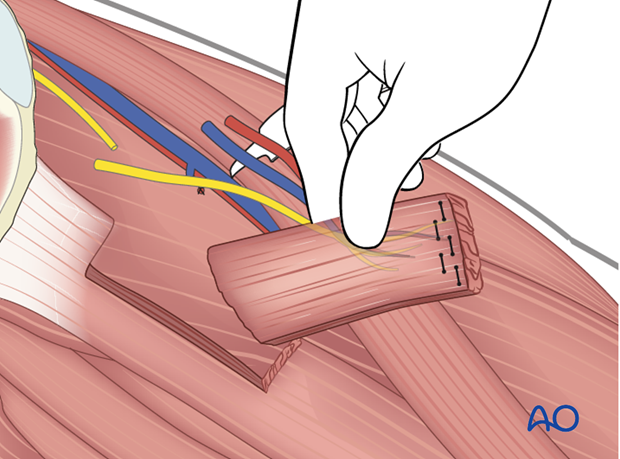
The obturator nerve and the neurovascular bundle are identified and dissected proximally to maximize length.
Muscle harvest
Generally, the anterior 50% and approximately 10 cm length of the gracilis is harvested, centered over the pedicle and the nerve as they enter into the muscle.
This point can be variable between patients.
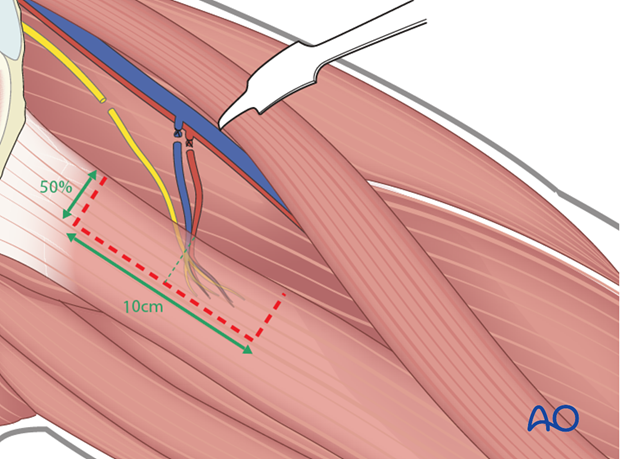
The exact amount and location of muscle harvested is determined by intraoperative stimulation of the obturator nerve fascicular bundles (usually 2-3).
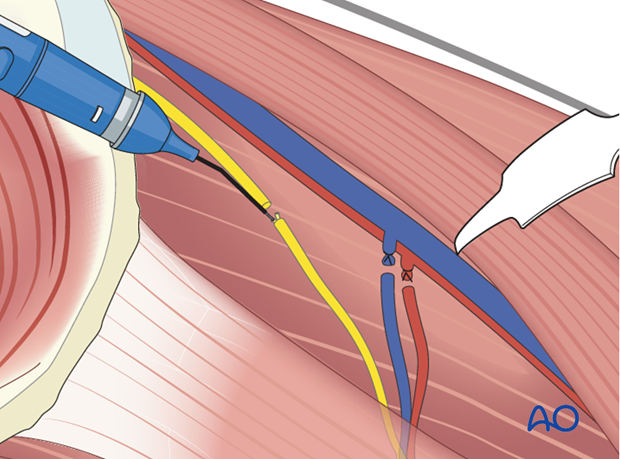
The muscle segment of interest is detached from the remaining muscle. Several permanent horizontal mattress sutures are tied in an overlapping fashion at the distal end of the muscle segment.
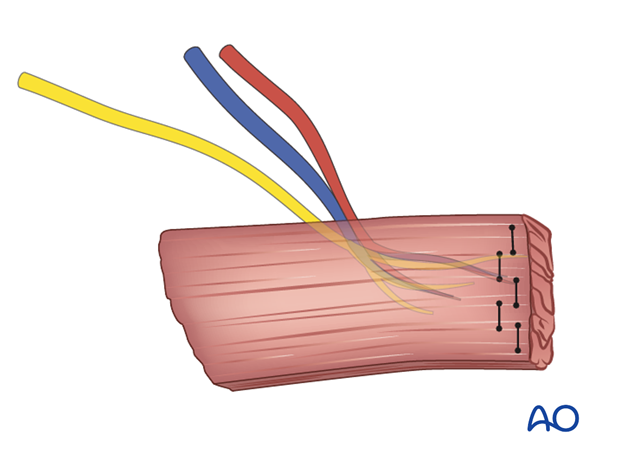
The muscle segment is harvested with its neurovascular bundle.
The donor site is closed in multiple layers over a closed suction drain.
3. Inset
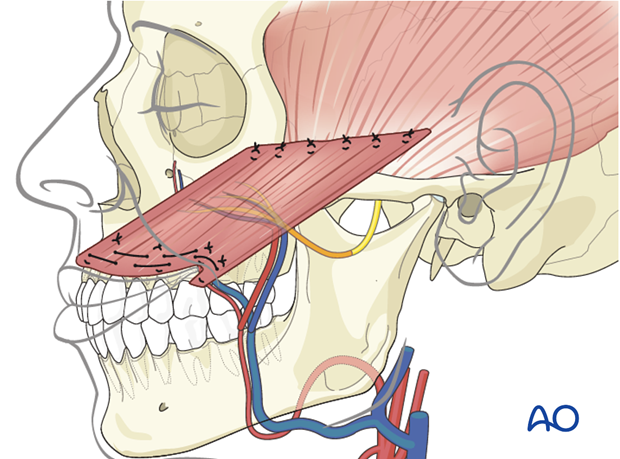
The objective of positioning the gracilis muscle is to simulate the vector of pull of the zygomaticus major muscle.
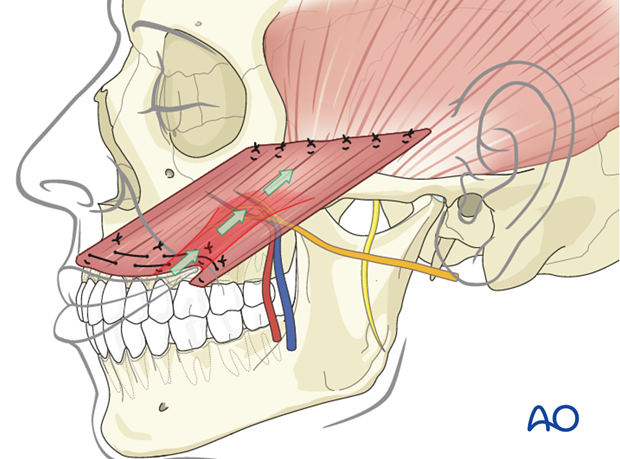
Flap orientation
The muscle flap is oriented with the neurovascular pedicle in the inferolateral position and on the deep surface, to optimize the geometry of neurovascular anastomosis. This orientation ensures that the reinforcing sutured distal end of the muscle is turned towards the mouth.
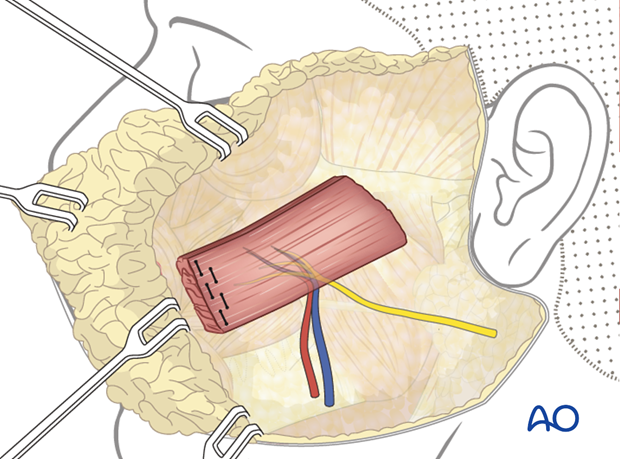
Flap fixation
Four fixation points centered around the commissure are identified in the following order:
- Oral commissure
- Mid lower lateral lip
- High point of Cupid's bowl ipsilateral
- Mid upper lateral lip
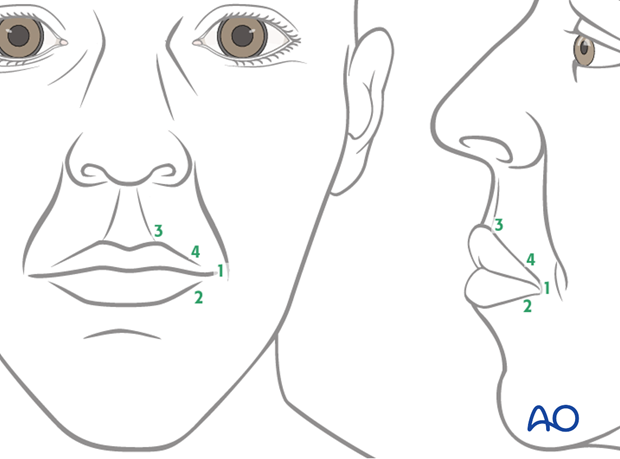
All points are identified through trial and error, using anchoring sutures and visual inspection. The goal is to achieve a natural smile vector and recreation of the nasolabial fold.
The muscle is anchored to the four fixation points using permanent horizontal mattress sutures.
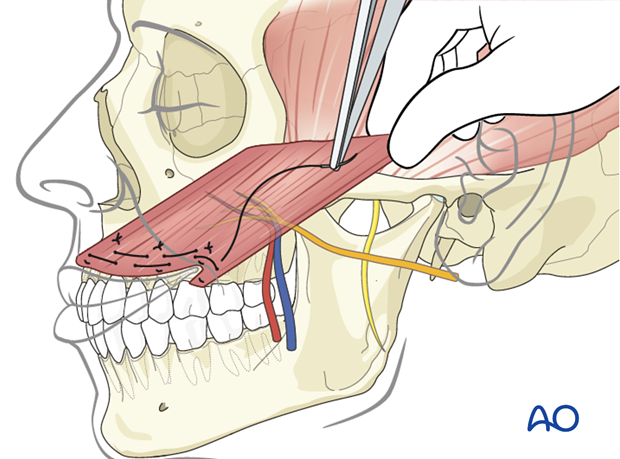
The muscle is placed under tension to achieve slight overcorrection of the smile; its proximal end is anchored medially onto the zygomatic body periosteum and laterally to the temporal fascia using permanent horizontal mattress sutures.
During anchoring the muscle at its proximal end is trimmed as required to achieve appropriate positioning.

The obturator nerve is coaptated to the masseteric nerve and the vessels are anastomosed to the facial artery and vein.
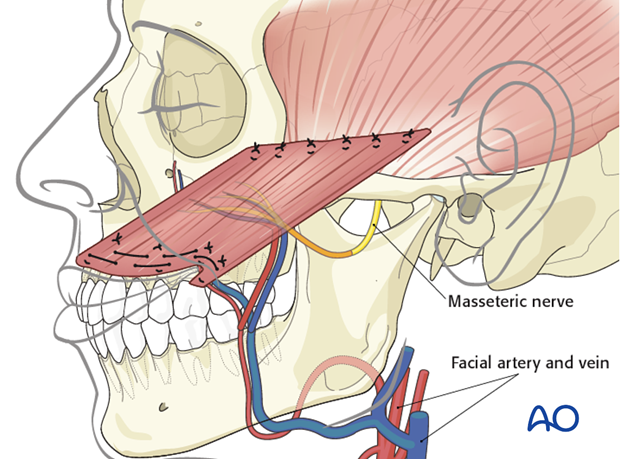
Closure
Standard closure in layers over an open drain is performed.
4. Case example: complete facial paralysis after skull base surgery
32-year-old male with complete irreversible left facial paralysis after skull base surgery.
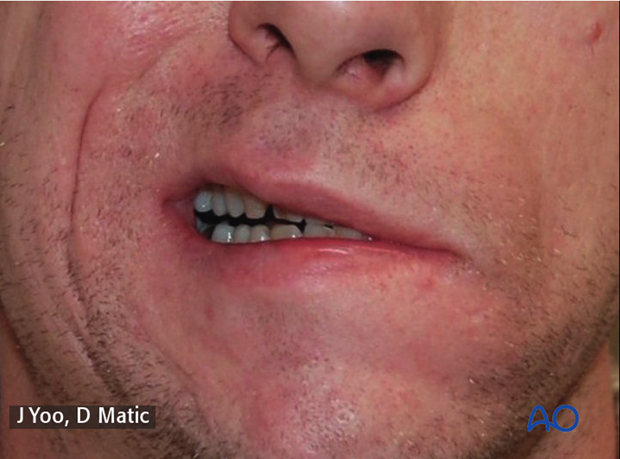
a) 4 months postoperative following gracilis innervated by masseteric nerve, gold weight implant, and lower eyelid tendon suspension.
b) Note improvement over time with biofeedback and relaxation of patient’s right (normal) side.
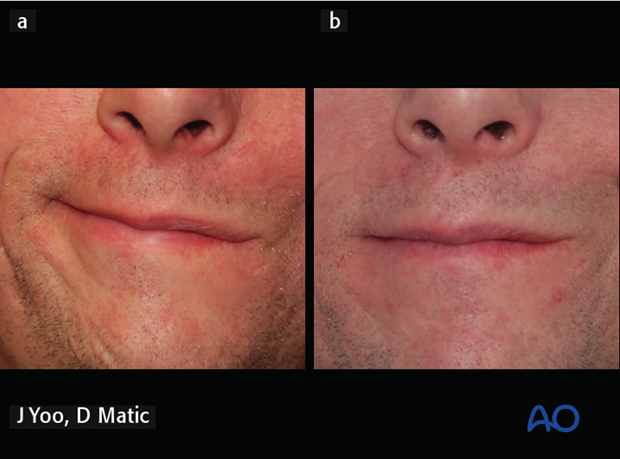
5. Case example: complete facial paralysis after skull base surgery
54-year-old female irreversible complete right facial paralysis after skull base surgery.
8 years postoperative, following gracilis innervated by masseteric nerve, static suspension with plantaris tendon for upper nasolabial fold, lower eyelid tendon suspension, gold weight implant.
The picture is showing the patient at rest and smiling.
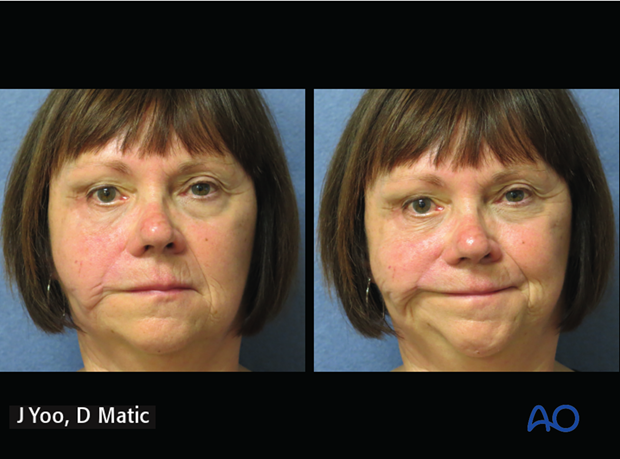
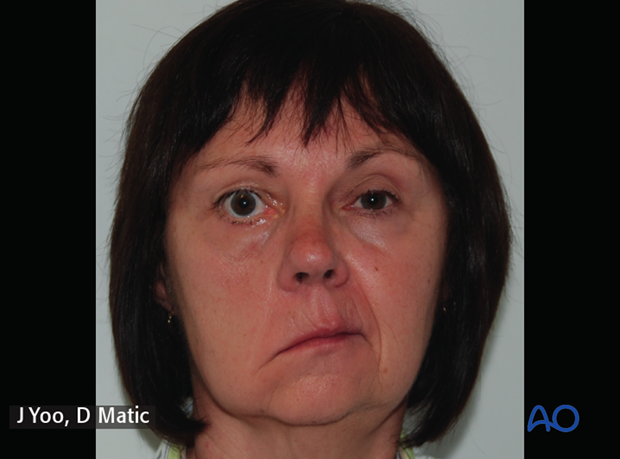
6. Case example: congenital complete facial paralysis
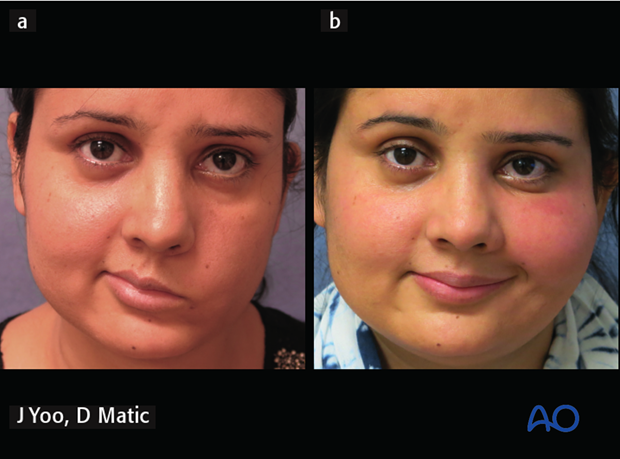
a) Preoperative picture of a 29-year-old female with congenital complete left facial paralysis.
b) 1-year postoperative following gracilis innervated by masseteric nerve.
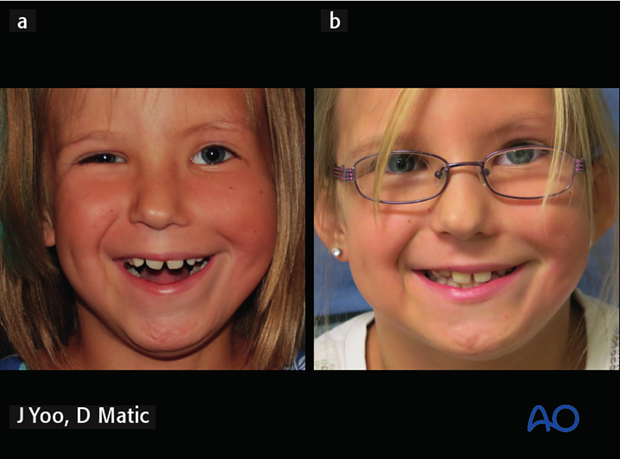
7. Case example: Moebius syndrome with complete facial paralysis
5-year-old female with Moebius syndrome with complete left facial paralysis.

a) 2 years following gracilis innervated by masseteric nerve.
b) 6 years postoperative - over time patient was able to smile without clenching (smile with mouth open).
8. Aftercare
The need for aftercare is to reduce the chance of inadvertent release of the suspension procedure. This can include maintaining soft diet 2-3 weeks postoperatively and minimizing exertion and trauma to the area during this period.
Generally, 4-6 weeks postoperatively patients are encouraged to return to normal activities without restrictions.
Infrequently, some patients may need formal mouth opening exercise if they develop trismus.
Specific physiotherapy exercises are started after this time. The goals are to improve symmetry both at rest and during function, through biofeedback and patient practice (in front of a mirror).
Postoperative free flap management
When a free flap is utilized, it should be regularly monitored to ensure vascular integrity. Hand-held Doppler or indwelling probes can be used to assess blood flow. In case of doubt of the vitality of the flap, surgical exploration may be necessary.
Aftercare of the donor site
Routine post-surgical wound care is required. A closed-suction drain is placed at the time of surgery and removed when minimal output (usually day 3). Patients ambulate and return to normal function without the need for directed physiotherapy.













