Anterior approach to the lumbar spine
1. Positioning for anterior procedures
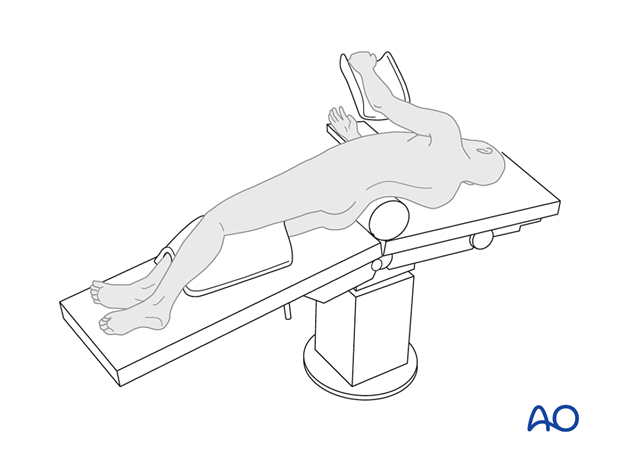
The patient is placed onto a radiolucent table and turned into the lateral decubitus position. Placing the patient on a surgical bean bag positioner is optional.
Padding is placed around both knees to prevent nerve damage.
The arm is elevated and placed in a well-padded support.
2. Anaesthesia
General anaesthesia with endotracheal intubation is required.
Anaesthesia maintenance should interfere minimally with spinal cord monitoring.
A propofol-based intravenous or inhalational technique using low minimum alveolar concentration (MAC) of isoflurane or sevoflurane (0.7 or less) with a remifentanil infusion is generally chosen.
High concentrations of nitrous oxide and inhalational agents interfere with spinal cord evoked potential monitoring.
When using motor-evoked potentials, muscle relaxants should be avoided.
Techniques to minimize blood transfusion during scoliosis surgery include avoiding hypothermia, controlled hypotension, intraoperative cell salvage and pharmacological agents such as tranexamic acid.
3. Blood management
Hypotensive anaesthesia (Mean arterial pressure (MAP) of 60 – 70 mmHg should be used during the exposure. Normotensive anaesthesia is recommended during the correction procedure to optimize blood flow to the spinal cord.
The use of a blood salvage techniques (eg. cell saver) is recommended.
Anti fibrinolytics (eg. tranexaminic acid or aminocaproic acid) can significantly reduce blood loss.
4. Preoperative antibiotics
Antibiotics should be administered well prior to the incision and also at 6h intervals or when the blood loss exceeds 2L.
A cephalosporin antibiotic with good gram positive coverage is generally recommended. Local bacterial spectrum will need to be taken into account, this should be discussed with the hospital microbiologist.
5. Spinal cord monitoring
Spinal cord monitoring is implemented. The risk of spinal cord injury during anterior lumbar (cauda equina level) scoliosis correction is significantly lower than for posterior surgery. Spinal cord monitoring may not be needed in all anterior lumbar scoliosis surgeries.
To monitor the integrity of the spinal cord and cauda equina intraoperative neuromonitoring should be performed.
Motor Evoked Potentials (MEP) and Somatosensory Evoked Potentials (SSEP) are optimal methods of intra-operative spinal cord monitoring.
In case of critical changes in the evoked potentials, the possibility of a wake up test needs to be available during the procedure.
In the event of signal changes, the following steps should be considered:
- Rule out equipment malfunction
- Surgical pause to reassess operative environment
- Alert anesthesiologist and surgeon
- Optimize anaesthetic agents, and patients temperature, and hemodynamic status
- Elevate blood pressure
- Reversal of any recently performed corrective maneuvers
- Rule out implant malposition
- Consider wake up test
- Consider steroid administration
- Consider radiographic evaluation
- Consider aborting procedure while providing spinal stability
6. Approach
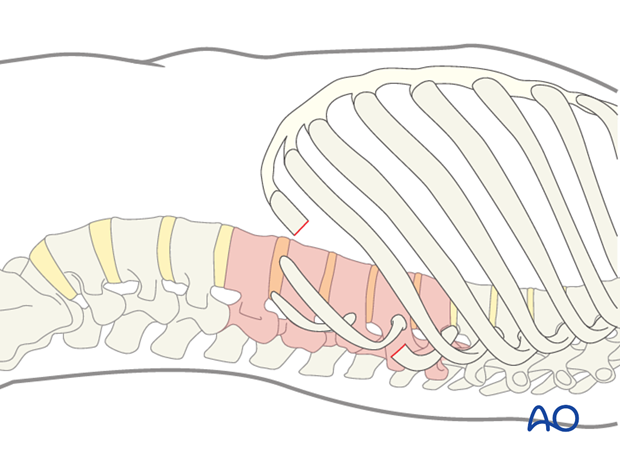
The incision is centered over the apex of the lumbar curve. This can be verified by image intensifier.
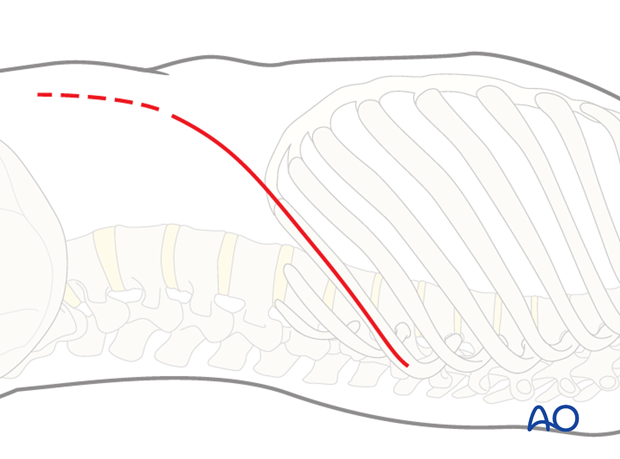
In general, the incision is made over a rib. Usually the rib that attaches one or two vertebrae superior to the upper instrumented vertebra (UIV) will be resected. Usually for a Lenke 5 and 6 curves the 10th, 11th or 12th rib will need to be removed.
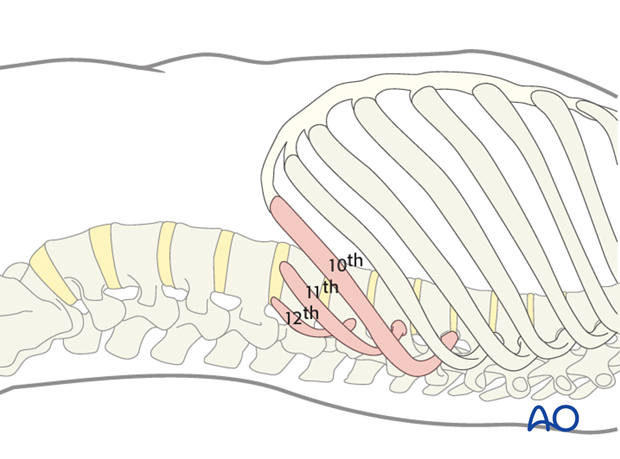
With this exposure 5 or 6 vertebrae can be readily instrumented.
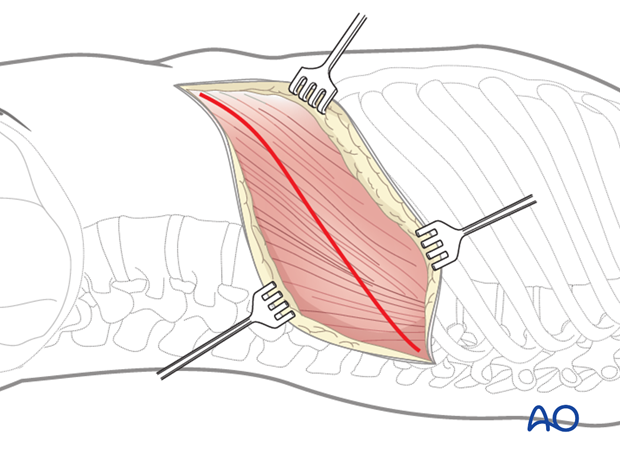
The chest muscles are incised with cautery, the latissimus dorsi can usually be spared by retracting it posteriorly.
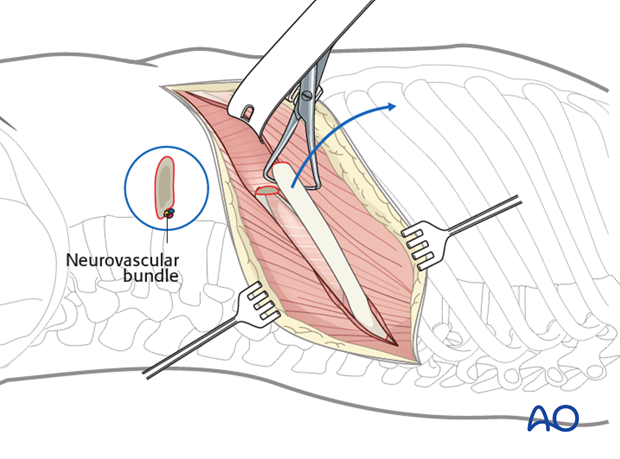
The rib is exposed subperiosteally, and 15 – 20 cm is resected.
Care is taken not to injure the neurovascular bundle which lies at the caudal edge of the rib.
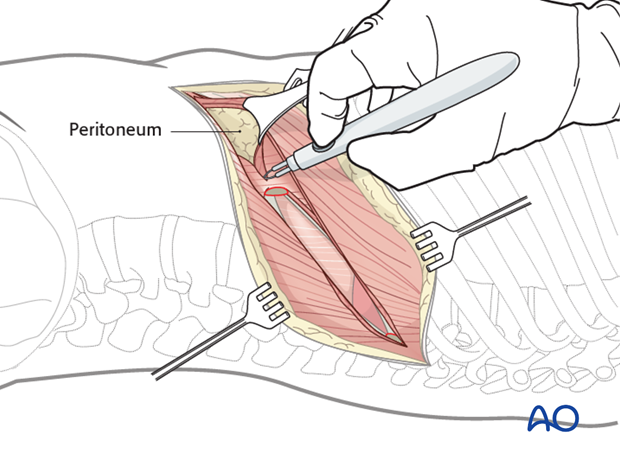
At the posterior part of the incision, the pleura parietalis is opened and the lungs exposed.
Further anterior the diaphragm attaches to the chest wall.

At the anterior part of the incision the three muscle layers of the abdominal wall are split, and the retroperitoneal cavity is exposed
After sweeping the peritoneum off the inferior surface of the diaphragm, it is taken down from the chest wall, all the way to the spine (down to the crus) leaving a 1 cm margin around the periphery for later reattachment. It may be useful to leave marking sutures every 3-4 cm to aid later reattachment.

The innervation of the diaphragm comes from the phrenic nerve near the esophagus, and any muscle left standing in the periphery will be denervated. Therefore it is important to cut the diaphragm close to the chest wall.
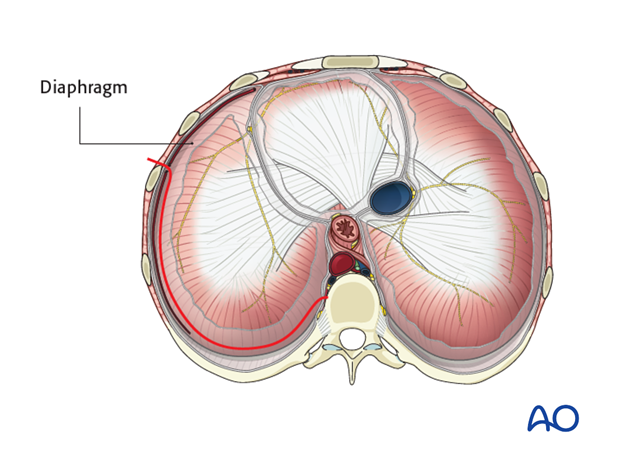
The diaphragm is then retracted anteriorly to join the retroperitoneal space and the intrapleural cavity.
Below L1 the peritoneum is retracted anteriorly and medially and the psoas muscle is exposed.
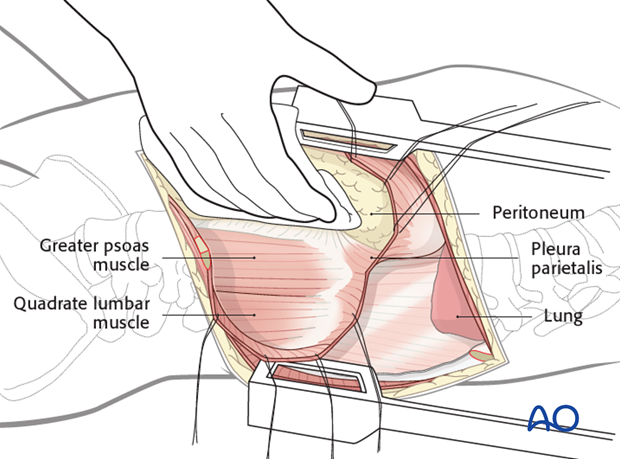
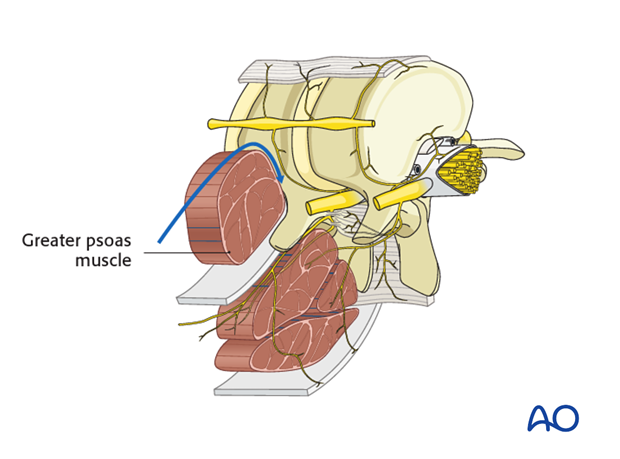
The psoas muscle is retracted posteriorly, partially detached from the spine using cautery, at each level making sure to identify the segmental vessels as well as the sympathetic chain, while avoiding entering into the neuroforamen.
Above L1 the pleura parietalis is cut longitudinally along the lateral aspect of the spine as far proximally as the UIV.
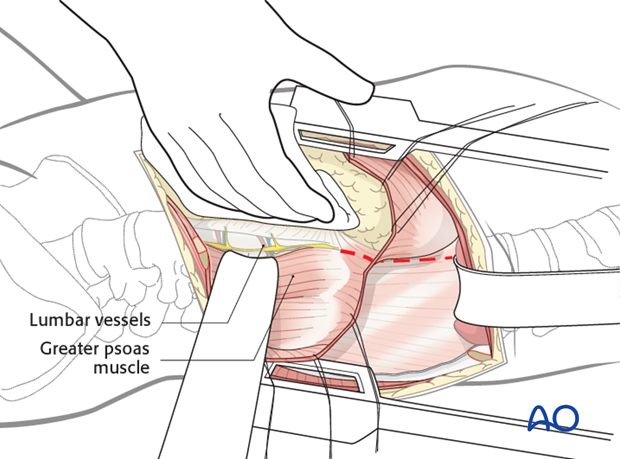
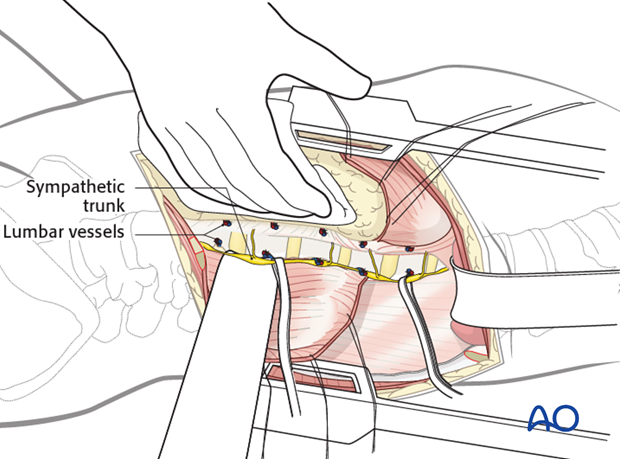
The segmental vessels that run over the vertebral bodies are exposed and ligated.
The sympathetic chain is bluntly retracted.
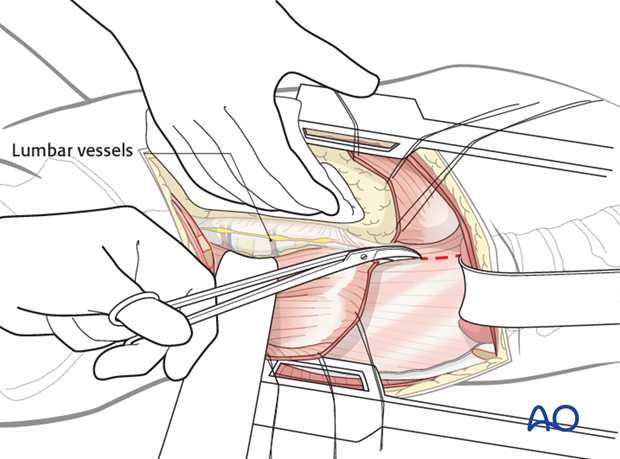
The spine is now exposed. Blunt retractors and swabs are placed anteriorly to protect the aorta and caval vein.
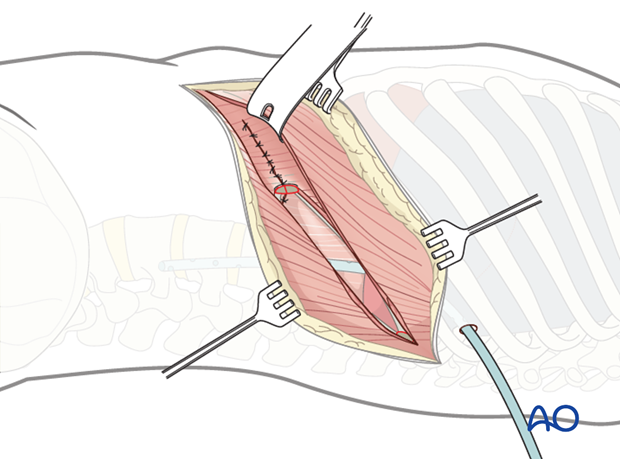
7. Closure
One or two chest tubes must be introduced in the mid-axillary line (some surgeons prefer an anterior drain to extract air from the chest cavity and a posteriorly placed drain for drainage of blood and fluid, other prefers just a single drain).
A retroperitoneal suction drain is optional.
The diaphragm is reattached using interrupted sutures, making sure that the pleura parietalis is approximated and closed preferably over the instrumentation.
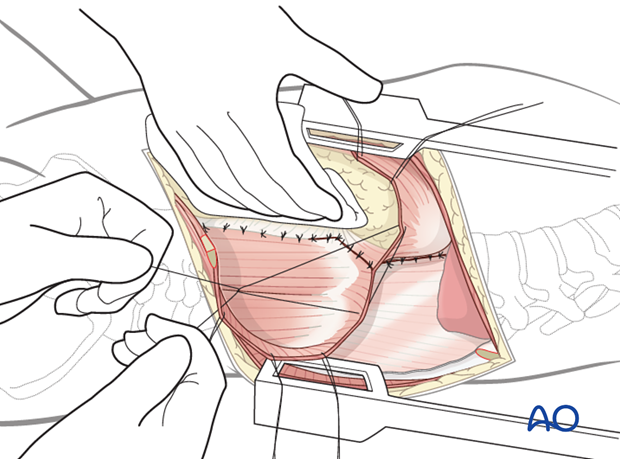
The three abdominal muscle layers are approximated.
The ribs are approximated using strong non resorbable sutures.
Chest musculature is reattached.
The subcutaneous tissues and skin are sutured in a layered fashion.