Diagnosis of wound infection
1. Diagnosis of wound infection
Introduction
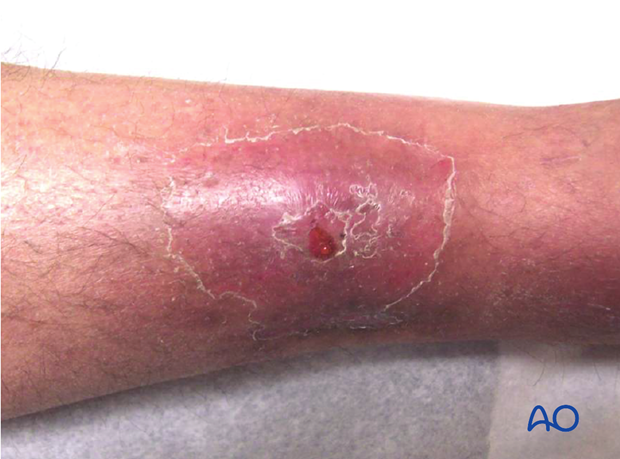
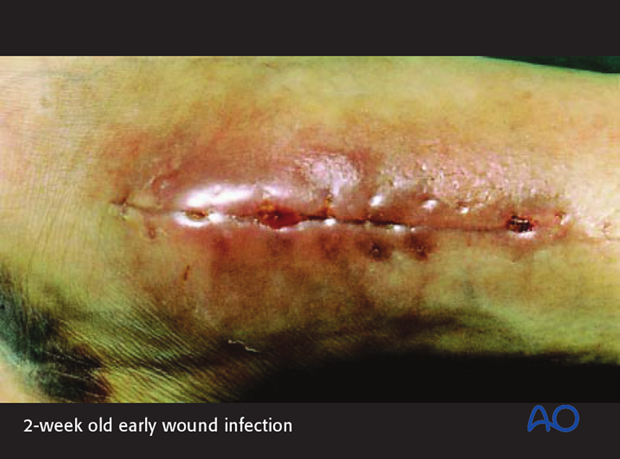
Early identification of a wound infection is the first step towards prompt treatment, which itself is a requirement for optimal results. Any fracture wound, whether due to the injury, or created by the surgeon, is at risk of becoming infected. Recognition of those factors which predispose to infection may increase the surgeon’s alertness to the possibility of infection. Early signs of infection are not specific and may easily be misinterpreted. Inflammation is normally present in the region of a fracture, even without infection. Often, the first sign of wound infection is that the inflammation fails to resolve normally. Certainly, increasing signs of inflammation (wound drainage, redness, swelling, pain and tenderness, fever) must be regarded as a strong indication of possible infection.
Should the surgeon be concerned about infection, efforts to diagnose, or exclude this possibility, must be undertaken without delay.
Evaluation of possible infection
Presence of bacteria within an inflammatory wound exudate is definitive proof of infection. Often this is most readily obtained by sterile aspiration of the wound, or by exploring it surgically. Microscopic examination (Gram’s stain) of the exudate, and appropriate bacteriologic cultures provide evidence of bacterial presence. Most fracture infections are due to readily evident bacteria, although previous antibiotic treatment may interfere with microbiology studies. It has to be borne in mind, additionally, that there may be few planktonic bacteria in the wound and that most bacteria may be trapped in biofilms. Occasionally, increasing inflammation is evident without recoverable bacteria. While occult infection may be the cause, the surgeon should remember that a non-infectious cause of increasing inflammation is mechanical failure of fracture fixation. This may also co-exist with infection.
Systemic signs of inflammation, often associated with infection, are provided by several laboratory studies (see below). By themselves, none of these prove nor exclude infection.
- Serial wound examinations
- Maximal daily temperature
- Complete blood count (CBC) with differential white cell count
- Erythrocyte sedimentation rate (ESR)
- C-reactive protein (CRP)
- Imaging
- Plain x-rays
- CT scan
- MRI
- Bone scan
- Bacteriology (Gram’s stain, culture and sensitivity)
Classification of infection
An important guide to treatment is provided by classifying an infection according to several parameters:
- Duration of infection
- Anatomical location
- Status of fixation
- Infecting organism(s)
- Viability of bone and soft tissue
- Host resistance to infection
Optimal management of an infected fracture requires consideration of each of these factors. Thus, each must be evaluated.
Clinical exam
Clinical symptoms and signs are most important for identifying the presence of infection. Increasing pain, drainage (either purulent or sero-sanguinous), swelling, redness, warmth and tenderness all suggest the possibility of a wound infection. Progressive worsening of one or more of these findings is confirmatory. Thus, serial examinations may be required. While some inflammation is caused by a fracture, its severity should progressively decrease. Local infection must not be mistaken for normal postoperative inflammatory signs.
Radiographic investigations
In early infections, x-ray imaging procedures typically play a minor role. Radiographic findings of infection are usually not evident until at least 2 weeks after the onset of infection, even though bone involvement has already occurred.
Imaging becomes important in the later stages of infection. It is helpful to examine serial x-rays for progressive changes that suggest infection.
Radiographic signs are neither sensitive nor specific for infection. E.g., radiographic evidence of implant loosening may be present with instability, infection, or both.
Ultrasound is useful to identify accumulation of fluid (hidden abscess). The method is non-invasive, and may reach deeper layers, especially in the thigh. Ultrasound can be helpful for guiding diagnostic needle aspiration.
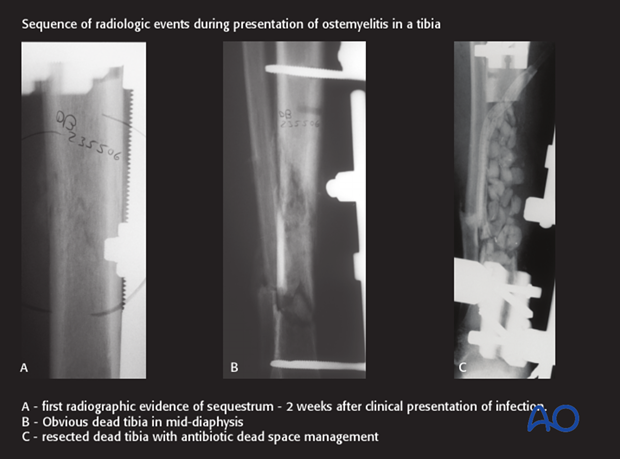
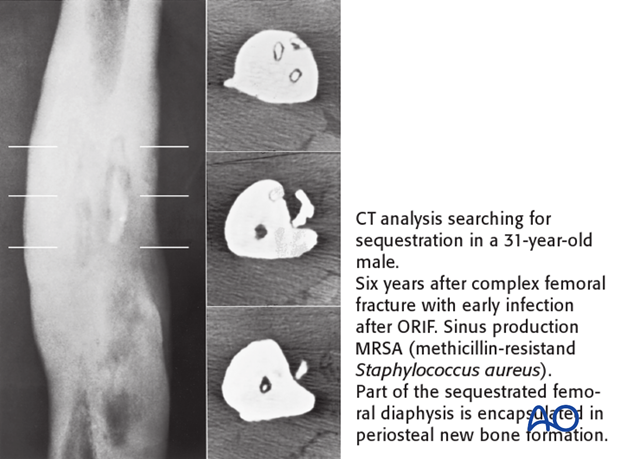
CT
Computed tomography better demonstrates differences in bone density, such as the dense white necrotic sequestrum illustrated here. It also offers a cross-sectional guide for exploration and debridement, particularly for bone fragments.
As with plain radiographs, CT scans offer no specific diagnostic sign for, or against, infection.
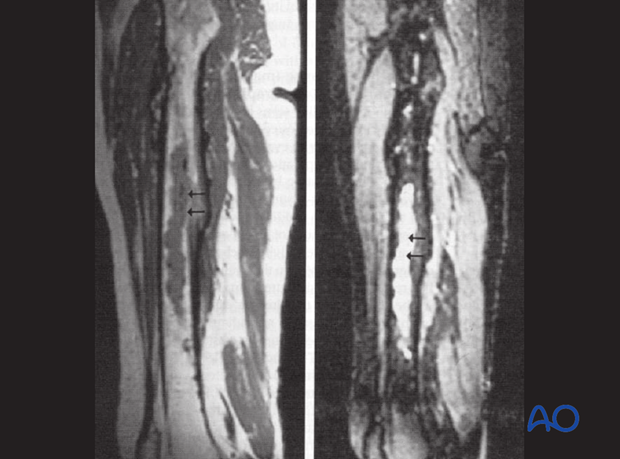
MRI
MRI presents an improved resolution of soft-tissue abnormalities and shows greater anatomic detail than other imaging studies. Once again, MRI signs of infection are non-specific. An additional disadvantage of MRI is the artifacts related to metal implants.
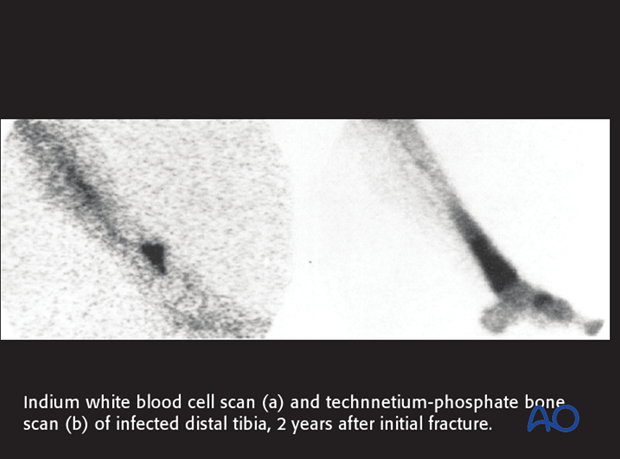
Bone scan
Uptake of technetium-labelled phosphate compounds (TCN-MDP) is increased in areas of high vascularity, including infections and bone healing. With infection, a 3-phase bone scan shows increased uptake of labelling in all 3 scan phases.
Absence of uptake suggests impaired vascularity, or bone necrosis. Bone scanning detects increased bone remodeling which is present around all fractures for 12-24 months. Bone scanning can not differentiate aseptic hardware loosening from infection. Bone scans have little value in the early postoperative period of acute fractures.
Indium-labelled white blood cell scans are more specific for inflammation and infection. The illustration shows a larger area of labelling (surrounding hyperemia and inflammation with TCM-MDP than with indium white blood cell technique). However, false positives and false negatives still occur, and such scans can be positive in un-united fractures.
Bacteriology
Fluid can be aspirated sterily preoperatively and sent for culture and Gram’s stain. Tissue specimens should be sampled from 3 or more suspicious sites, with two pieces of tissue from each site (one for microbiology and one for histopathology). Both aerobic and anaerobic cultures should be undertaken.
PCR bacterial identification, if available, is a more rapid and reliable technique than standard cultures.
Histological investigation can reveal a bacteriological etiology even if the bacteriological tests are negative. Superficial wound swabs should be avoided because of low sensitivity and frequent contamination by bacteria not responsible for the infection. Prior to tissue-sampling for culture, it is important to discontinue any antibiotic therapy for at least a week, or two.
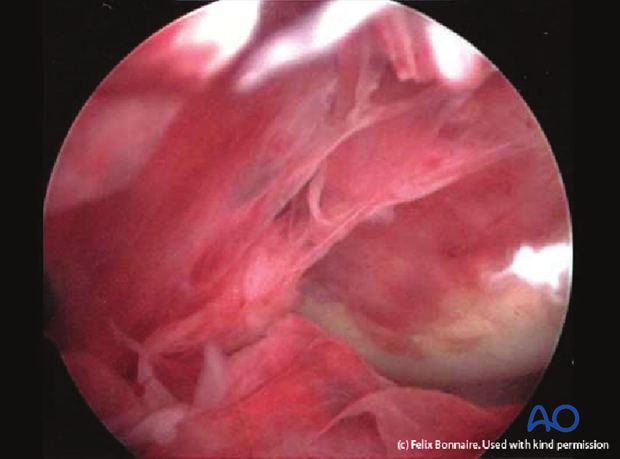
Septic arthritis
If the infection possibly involves a joint cavity, the need for draining and debriding that space must be appreciated. At the slightest suspicion of septic arthritis, joint aspiration should be performed to evaluate the affected joint. If there is presence of infected fibrinous deposits, arthroscopic, or open synovectomy must be performed. Arthroscopic irrigation should be performed repeatedly, every 2-3 days, until inflammation resolves. If arthroscopic debridement is unsuccessful, consider open synovectomy. If there is cartilage degradation, arthrodesis may be unavoidable. Infected joint wounds are different from hematogenous septic arthritis, and usually require surgical treatment.